 Chemistry
Chemistry
4665872 / 5558097
Noble Gases
A Family of Noble Disposition
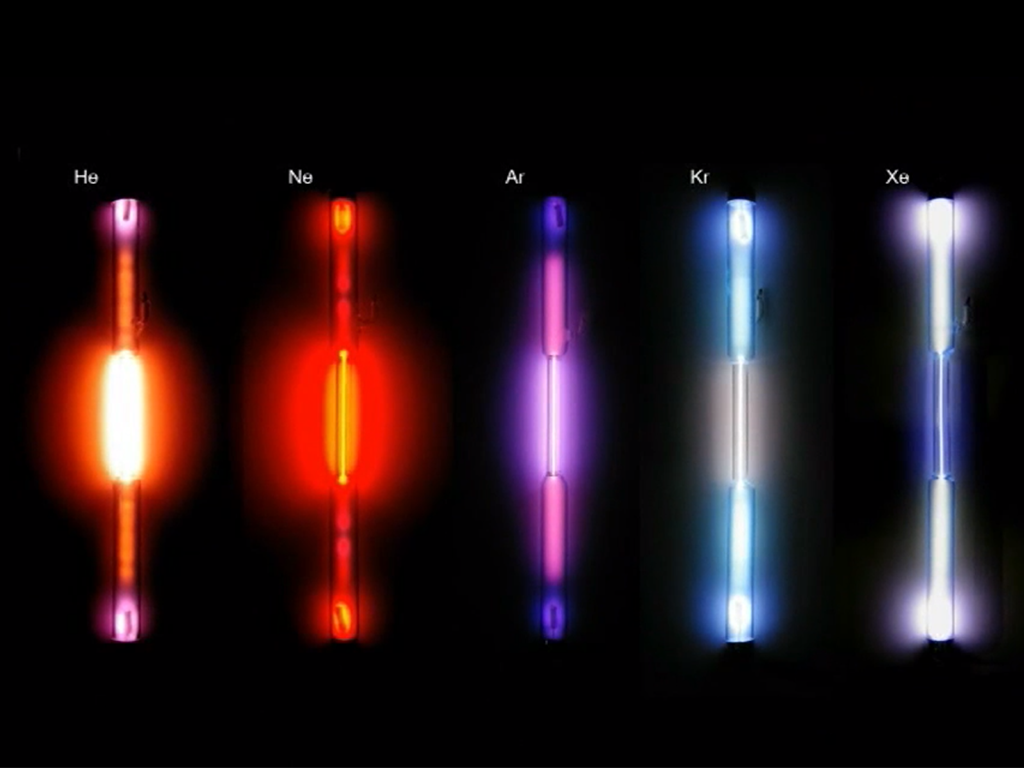
Xenon, Helium, Neon, Argon, Krypton and the radioactive Radon belong to the noble gases. These form the family of noble gases as the elements of the eighth group of the periodic table. All of them are colourless and odourless, non-inflammable and non-toxic. Their most striking chemical property is their inertness. This can be explained by their electron arrangement, termed noble gas configuration and represents a particularly stable and therefore low-energy state. The noble gases are to be found in scant amounts in our air from which they are also distilled. Helium is mainly extracted from natural gas. In everyday life, we encounter noble gases for example as shielding, filling or buoyant gases and in fluorescent tubes. The shell model describes the structure of the atoms. It is based on the distribution of electrons in restricted areas at a fixed distance around the core of the atom.
Play trailer
Curriculum-centred and oriented towards educational standards
Matching
Blogging
The weblog or blog, for short, as a medium is not much older than this century. Blogs came into being in the World Wide Web as ’messages from below’, as web pages from web creators who wanted to share their view of the world with the world. They are short notes, long texts, pictures, videos, which are posted loosely and at random intervals to the world for an undefined public.
Youth Movement
Dancing until your feet hurt: Here, at the meeting on the Hoher Meissner near Kassel, 3,500 participants from Boy Scout associations, youth and Wandervogel groups from all over the German-speaking region have gathered. They want to celebrate, simply get to know each other and commemorate a historic anniversary.









