Suche:
- # Artistry
- # Biology
- # Chemistry
- # Ecological
- # Economy
- # English
- # Foreign Language
- # Geography
- # German
- # Health
- # History
- # Informatik
- # Latin
- # Mathematics
- # Media Education
- # Music
- # Physics
- # Politics / Civics
- # Preschool
- # Primary School
- # Religion
- # Society
- # Sports
- # Technology
- # Training of Teachers
- # Vocational Education
Feuer und Flamme
Zehn interaktive H5P-Aufgaben, die den Ablauf einer Schulstunde nachvollziehen, erklären in Videos Verbrennungsreaktionen und sichern das Wissen mit Aufgaben ab.
Learn moreSaure und alkalische Lösungen im Alltag
Zehn interaktive H5P-Aufgaben, die den Ablauf einer Schulstunde nachvollziehen, erklären in Videos saure und alkalische Lösungen und sichern das Wissen mit Aufgaben ab.
Learn moreLuft und ihre Zusammensetzung
Zehn interaktive H5P-Aufgaben, die den Ablauf einer Schulstunde nachvollziehen, erklären in Videos die Zusammensetzung der Luft und sichern das Wissen mit Aufgaben ab.
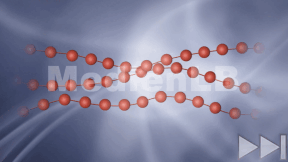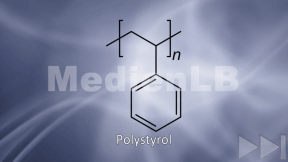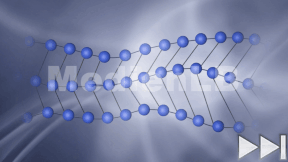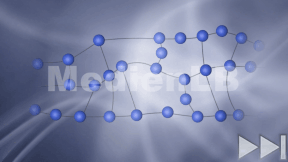
Learn moreKunststoffe
Zehn interaktive H5P-Aufgaben, die den Ablauf einer Schulstunde nachvollziehen, erklären in Videos Kunststoffe und sichern das Wissen mit Aufgaben ab.
Learn moreIsotope
Zehn interaktive H5P-Aufgaben, die den Ablauf einer Schulstunde nachvollziehen, erklären in Videos Atomaufbau und Isotope und sichern das Wissen mit Aufgaben ab.
Learn moreDünnschichtchromatographie
Chromatografische Verfahren sind physikalische / chemische Trenntechniken von Stoffen.
Learn moreLerneinheit Chemie 8 – Kalk
In unserem Arbeitsheft „Lerneinheit Chemie 8 – Kalk“ finden Sie 10 interaktive und didaktisch aufbereitete Aufgaben zum Thema Kalk.
Aluminium II
The metal aluminium is growing in importance because of its specific properties and manifold application possibilities. This DVD deals with the industrial production of aluminium as a raw material, its processing and the manufacturing of alloys for the finished product. Starting with the raw material aluminium oxide the functioning of an electrolytic cell is demonstrated and explained. Alumina, white and powdery, is melted with great expenditure of energy, and by means of electrolysis converted into aluminium with a degree of purity of 99.9%. As aluminium oxide would not melt before a temperature of over 2,000°C is reached, the mineral cryolite is used as a solvent. The various alloys change the properties of aluminium and are produced according to precise formulations. The alloy is cast into blocks and bars that serve as primary material for processing plants. The responsible handling of resources underscores the importance of recycling. Aluminium is resilient and versatile.
Learn moreAluminium I
In the modern world, we encounter aluminium at every turn. This is due to the particular properties of the metal. Increasingly, aluminium is about to edge iron and steel out of engineering, as aluminium allows energy-saving lightweight construction of aircraft and vehicles of all kind. Aluminium is weather-resistant, does not rust and is therefore well suited as building material for house facades, window frames or simply for all parts that are exposed to wind and weather. At the same time, aluminium has a noble-looking surface recommending it as material for interior design.
Learn morePlastic
Plastic has been around for not longer than roughly 100 years, and the synthetic material is a brilliant invention. Its production is cheap, it can take almost any possible form, it is light-weight, versatile and, above all, inexpensive.
Learn moreC, CO2 and Associates in Everyday Life
All organic matter contains carbon. Coal is deposited in the Earth's interior. It developed about 300 million years ago from plants in a geological period which is also called Carboniferous. During the combustion of organic matter, carbon turns into the gas carbon dioxide. Dissolved in water, it becomes the so-called carbonic acid. Carbon dioxide is an incombustible, colourless and odourless gas that is easily dissolved in water. With various metal oxides or hydroxides it forms two types of salts: the carbonates and the hydrogen carbonates. As calcium carbonate it is contained in natural products such as chalk and egg shells. Specific forms of carbon, called modifications, are graphite and also the particularly valuable diamond.
Learn more