55502493
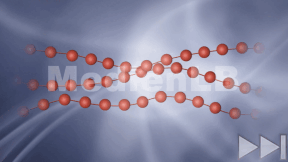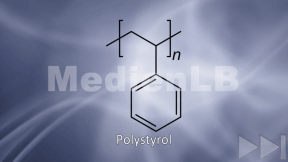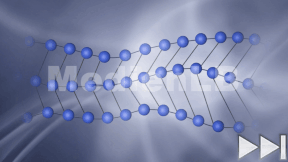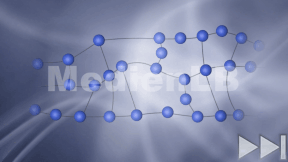
Kohlenstoff
In 10 interaktiven Modulen und in interaktiven Videos wird Wissen zu Kohlenstoff vermittelt und abgefragt.
Das Medium bietet H5P-Aufgaben an, die ohne zusätzliche Software verwendbar sind.
Durch interaktive Aufgabentypen wird das audiovisuelle und interaktive Lernen einfach.
Lernen macht jetzt Spaß!
Included Tasks
- I Was ist Kohlenstoff - Lückentext
- II Kohlenstoff im Alltag - Aufgaben mit Video
- III Kohlenstoffarten - interaktives Video
- IV Kohle und Kohlenstoff - interaktives Quiz
- V Kohlenstoff im Alltag - interaktive Aufgabe
- VI Summenformeln für Spezialisten - interaktive Flashkarten
- VII Strukturformeln und Summenformeln - interaktive Aufgabe
- VIII Kohlenstoff im Buchstabengitter - interaktive Aufgabe
- IX Säure-Basen-Eigenschaften - interaktive Aufgabe
- X Kohlenstoff- interaktive Aufgaben
Curriculum-centred and oriented towards educational standards
Matching
Materials and Substances of Everyday Life
Hearing these words, you first think of the materials our clothing is made of. But all objects surrounding us in everyday life consist of one or several materials.
Plastic
Plastic has been around for not longer than roughly 100 years, and the synthetic material is a brilliant invention. Its production is cheap, it can take almost any possible form, it is light-weight, versatile and, above all, inexpensive.
Acids and Bases
We can find acids and bases in every supermarket, some of them in our food, others in cleaning agents. In everyday products, acids and bases as well as acidic and alkaline reacting salts have extremely different functions. In food, acids are either present or added as flavouring agents such as citric acid, tartaric acid and acetic acid, as antioxidants such as ascorbic acid or generally as acidifiers, sequestrants (citric acid and tartaric acid) and preservatives (acetic acid).