 Chemistry
Chemistry
4665872 / 5558097
Noble Gases
A Family of Noble Disposition
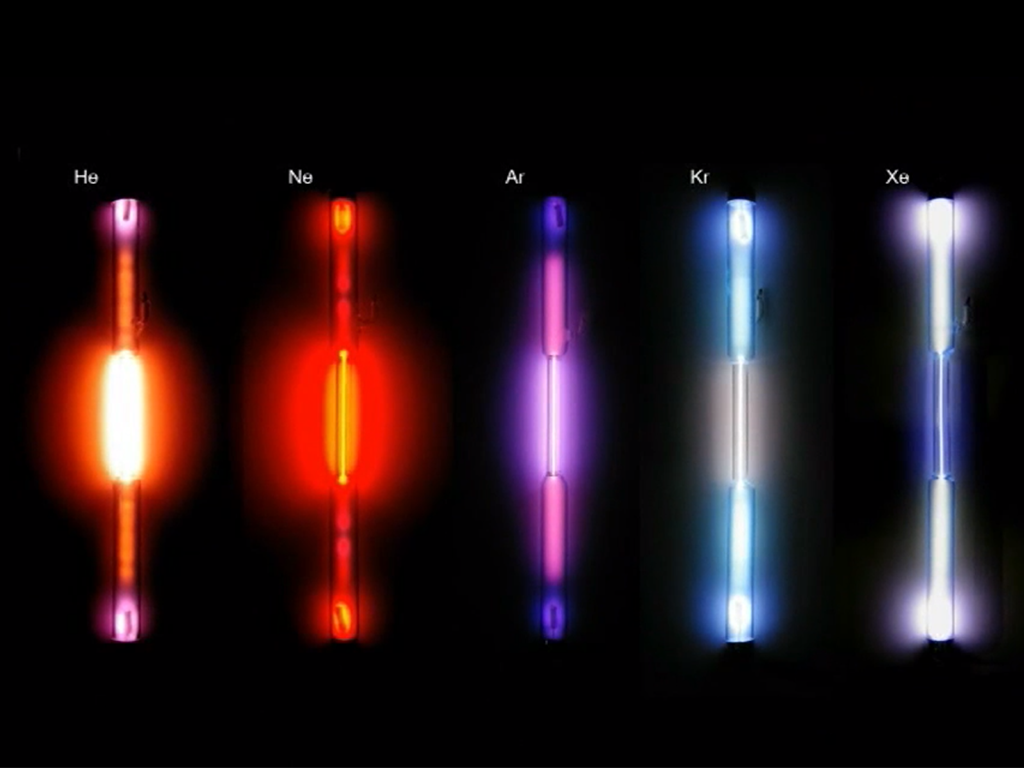
Xenon, Helium, Neon, Argon, Krypton and the radioactive Radon belong to the noble gases. These form the family of noble gases as the elements of the eighth group of the periodic table. All of them are colourless and odourless, non-inflammable and non-toxic. Their most striking chemical property is their inertness. This can be explained by their electron arrangement, termed noble gas configuration and represents a particularly stable and therefore low-energy state. The noble gases are to be found in scant amounts in our air from which they are also distilled. Helium is mainly extracted from natural gas. In everyday life, we encounter noble gases for example as shielding, filling or buoyant gases and in fluorescent tubes. The shell model describes the structure of the atoms. It is based on the distribution of electrons in restricted areas at a fixed distance around the core of the atom.
Play trailer
Curriculum-centred and oriented towards educational standards
Matching
Ceramic
Ceramics are indispensable in our everyday lives. We eat from ceramic plates, drink from ceramic cups, use tiled ceramic bathrooms. But how is ceramic manufactured? The film reveals the secrets of this fascinating material! We get to know more about the beginnings of ceramic in the Old World of Egypt and Mesopotamia, about Greece, China and Rome. We gain interesting insights into the valuable earthenware and are also shown the exquisite further development of the "white gold". Today this versatile material is irreplaceable in industry, too. Whether in space or as an easily compatible substitute in medicine, ceramic is applied in many places.
Inclusion
Madita is eleven and blind. She does not want to go to a special school but to a regular grammar school. She says she feels "normal" there. Jonathan is eight and has a walking disability. He likes going to the school where he lives. Here, his best friend sits next to him. Max Dimpflmeier, a teacher who is severely deaf, explains that school life is not easy. Quote Max Dimpflmeier: "You don't want to attract attention, you want to avoid saying that it is necessary for you that 70 people adjust to your situation." People on their way to inclusion.
Blogging
The weblog or blog, for short, as a medium is not much older than this century. Blogs came into being in the World Wide Web as ’messages from below’, as web pages from web creators who wanted to share their view of the world with the world. They are short notes, long texts, pictures, videos, which are posted loosely and at random intervals to the world for an undefined public.









